AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Sigma bond meaning12/5/2023  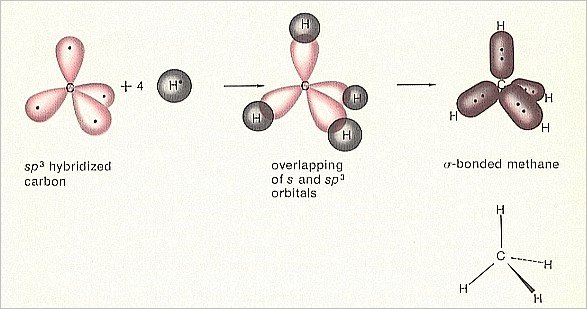
Bond Hybridization Practice (MOC Membership).Orbital Hybridization And Bond Strengths.How To Determine Hybridization: A Shortcut.Bond Dissociation Energies = Homolytic Cleavage.Five Key Factors That Influence Acidity.Question: Relative to sigma bonds, do you think p-p bonds (π bonds) would be stronger or weaker?Ģ) Energy required to break C-C bond in ethane:Įnergy required to break apart the C=C bond in ethene: In contrast to sigma orbitals, there is only one way to form a C-C π bond – from the overlap of two p orbitals. Now, the number of π bonds that can form will be dependent on the number of unhybridized p orbitals available – 1 for sp 2 hybridized carbons, 2 for sp hybridized carbons (the two π bonds will be at right angles to each other in the latter case). General principle – the more s character the bond has, the more tightly held the electrons will be. Take a look at the relative bond lengths and strengths for each of these six situations. The electrons in s orbitals are held more closely to the nucleus than electrons in p orbitals. Now, recall that for any given quantum number, s orbitals are lower in energy than p orbitals. Since carbon can exist in one of these three hybridization states, we can therefore have six varieties of carbon-carbon sigma bonds: Those orbitals will be some combination of s orbitals and p orbitals. Sigma bonds are created from the head-on overlap of orbitals. It is these hybrid orbitals that form sigma bonds (σ bonds). sp 2 = 33% s character, 66% p character.sp 3 = 25% s character, 75% p character.The “flavor” of each bond depends on the relative proportions of s orbital and p orbital content: If you draw an analogy to how we could make a “hybrid” soft drink by mixing different proportions of Sprite and Pepsi, these new orbitals aren’t fully s or fully p, but are a combination of both. What’s observed from analyzing the structure of molecules such as CH 4 is that the shapes cannot result from the electrons being in s or p orbitals alone, but instead are a consequence of the electrons in s and p orbitals mixing to form hybrid orbitals. Hybridization is a concept that we address here. The p orbitals therefore fill with electrons only after the s orbitals are filled. The electrons in p orbitals are slightly farther away from the nucleus than those in s orbitals, so they are a little bit higher in energy. P orbitals are shaped like figure-eights, or loops. 
The electrons of hydrogen, for instance, are in a 1s orbital. S orbitals should be familiar as the spherical-shaped orbitals. You may recall from Gen chem (and no doubt your first week of o-chem as well), that orbitals on carbon come in two flavors: s and p. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
